FDA VRBPAC Hopelessly Chasing Outgoing XBB.1.5 as Fall Target
Strain is 27% and Shrinking Fast--New Vaccine will be Obsolete on Arrival
By Peter A. McCullough, MD, MPH
From Rita Rubin, Science Reporter for JAMA: “On June 15, members of the US Food and Drug Administration’s (FDA) Vaccine and Related Biological Products Advisory Committee (VRBPAC) voted unanimously to recommend updating the COVID-19 vaccine composition to a monovalent XBB lineage.
On June 16, the FDA announced that it had advised manufacturers planning to update their COVID-19 vaccines that they should specifically target XBB.1.5. Scientists from Moderna, Novavax, and Pfizer had told the FDA and its advisory committee that their XBB.1.5 monovalent vaccines could be ready to inject into arms by late July or early fall.
Although the FDA decides what antigens the COVID-19 vaccines should include, the US Centers for Disease Control and Prevention (CDC) is responsible for deciding who should get them and when. As soon as the FDA greenlights an XBB.1.5 vaccine, “I’m sure the ACIP will have a specially called meeting” to decide how it should be used, William Schaffner, MD, chair of the department of preventive medicine at the Vanderbilt University School of Medicine, said in an interview.
ACIP stands for the CDC’s Advisory Committee on Immunization Practices, on which Schaffner serves as the liaison representing the National Foundation for Infectious Diseases, where he is medical director. At the ACIP meeting on an XBB.1.5 vaccine, “I think there will be a rather elaborate discussion on who will receive this vaccine,” Schaffner predicted.”

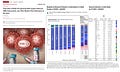
Shaffner and the VRBPAC should take a look at the CDC Nowcast System which shows XBB.1.5 at 27% of strains and falling fast. That means by the the fall the new monovalent XBB.1.5 vaccine would be completely obsolete. It appears that the FDA, CDC, and vaccine developers will never be able to get ahead of the now weakened but agile Omicron strains. They have still not addressed unacceptable safety data which overshadows discussions on “who should receive the product.”
Please subscribe as a paying or founder member so we can continue to bring you the truth.
CDC COVID Tracker Variant Proportions Monitoring Variant Proportions, Accessed July 5, 2023



You can't vaccinate against a coronavirus. It mutates too fast and too much. Doctors used to know that, but they're too stupid now.
I have been saying since the outset, that trying to create a vaccine for a corona virus, is akin to chasing the wind. And that would be true, even if these were actually vaccines, which they are clearly not. Further, there is overwhelming evidence that the mRNA injections are dangerous & deadly to an unprecedented degree. That our federal health agencies both ignore this trend & continue to double down on their promotion & administration speaks to their deliberate malfeasance. Since they now dictate to the corporate medical community, with serious reprisals for those who do not follow their guidelines, what we have is a combination of democide & iatrogenicide. For a stark look at how corporate administrators control their medical staff, I highly recommend the book, "If I Betray These Words", by Wendy Dean, MD.